Core Concepts
Genes ADRA2A and IRX1 linked to Raynaud disease.
Abstract
Researchers identified genes ADRA2A and IRX1 linked to Raynaud disease, impacting blood vessel constriction and dilation. The study, published in Nature Communications, analyzed data from the UK Biobank and Queen Mary University of London Genes & Health Study. Primary Raynaud is the focus, with potential implications for secondary Raynaud. New therapeutic avenues may arise from these genetic findings.
Researchers Link Two Genes to Raynaud Disease
Stats
Raynaud affects 2%-5% of the general population.
5100 individuals with Raynaud identified in the UK Biobank.
ADRA2A encodes for alpha-2A adrenergic receptor.
IRX1 gene associated with regulating blood vessel dilation.
Quotes
"Our finding of alpha-2A receptors is quite interesting because the focus has always been on alpha-2C receptors." - Maik Pietzner
"The study is the largest of this kind that's been done." - Laura Hummers
Key Insights Distilled From
by Lucy Hicks at www.medscape.com 10-19-2023
https://www.medscape.com/viewarticle/997555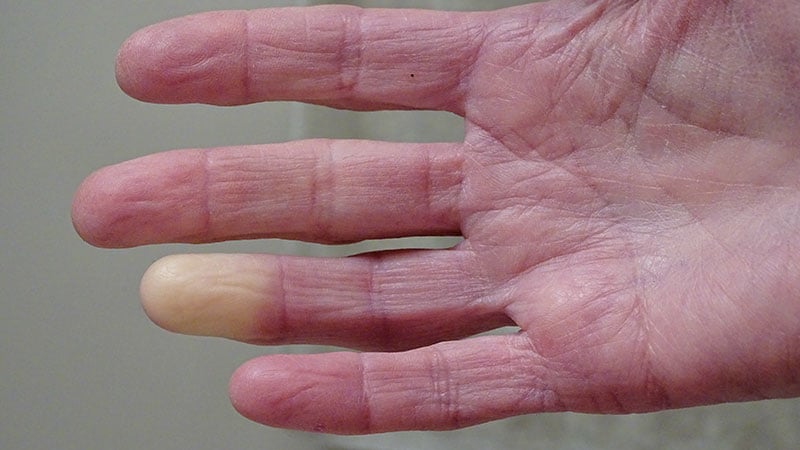
Deeper Inquiries
How might genetic findings impact the development of personalized treatments for Raynaud disease?
The genetic findings related to Raynaud disease, specifically the identification of genes like ADRA2A and IRX1, can significantly impact the development of personalized treatments. By understanding the genetic basis of the condition, researchers and clinicians can tailor treatments to target specific pathways or mechanisms involved in Raynaud. For instance, if a patient is found to have overexpression of the ADRA2A gene, which is associated with vasoconstriction, a personalized treatment approach could involve using selective inhibitors like mirtazapine to counteract this effect. This personalized approach based on genetic findings can lead to more effective and targeted therapies for individuals with Raynaud disease.
What are the potential implications of the study's focus on primary Raynaud for understanding secondary Raynaud?
While the study primarily focused on primary Raynaud, the implications of these findings can extend to understanding secondary Raynaud as well. Primary Raynaud is more common and often milder compared to secondary Raynaud, which is associated with autoimmune disorders like scleroderma and tends to be more severe. However, since the identified genes, such as ADRA2A and IRX1, are likely involved in the fundamental mechanisms of Raynaud, the insights gained from studying primary Raynaud can provide valuable information for understanding the pathophysiology of secondary Raynaud. This cross-over of knowledge can potentially lead to improved diagnostic and therapeutic strategies for individuals with secondary Raynaud.
How can the limitations of using electronic medical records in research be addressed to ensure accurate findings?
To address the limitations of using electronic medical records (EMRs) in research and ensure accurate findings, several strategies can be implemented. Firstly, researchers can validate the diagnosis of Raynaud by incorporating additional clinical assessments or diagnostic criteria beyond EMR data. This can help in confirming the accuracy of the identified cases and reducing the inclusion of misdiagnosed individuals. Secondly, efforts should be made to include a more diverse population in the study to enhance the generalizability of the findings. By expanding the demographic representation, researchers can mitigate biases associated with studying a homogenous group. Additionally, utilizing multiple data sources and cross-referencing EMR data with other sources of information can enhance the robustness of the study findings and minimize potential errors. By implementing these strategies, researchers can overcome the limitations of EMR analyses and ensure the reliability and validity of their research outcomes.
0
