Second Pig Heart Recipient Dies at University of Maryland Medical Center
Core Concepts
Man with terminal heart disease, Lawrence Faucette, who received a genetically modified pig heart transplant, passed away, highlighting the challenges and risks of xenotransplantation.
Abstract
Lawrence Faucette, a 58-year-old man with terminal heart disease, received a genetically modified pig heart transplant at the University of Maryland Medical Center (UMMC) after being turned down for a standard allograft transplantation. Despite initial progress, Faucette's heart showed signs of rejection, leading to his passing. The medical team aims to learn from this experience to improve future xenotransplant procedures.
Lawrence Faucette received a pig heart transplant on September 20, 2023, after being admitted to UMMC in end-stage heart failure.
The US FDA granted emergency authorization for the surgery through its single-patient investigational new drug compassionate use pathway.
Faucette expressed hope and gratitude for the opportunity the pig heart transplant provided him.
Despite initial progress and participation in physical therapy, Faucette's heart showed signs of rejection before his passing.
The medical team, led by Dr. Bartley P. Griffith, aims to learn from Faucette's case to improve future xenotransplant procedures.
Dr. Muhammad M. Mohiuddin highlighted Faucette's contribution to advancing the field of xenotransplantation and emphasized the importance of learning from each case.
UMMC performed the first genetically modified pig heart transplant in January 2022, with the recipient surviving for 60 days.
Second Pig Heart Recipient Dies
Stats
Lawrence Faucette received the pig heart transplant on September 20, 2023.
The first genetically modified pig heart transplant was performed in January 2022.
Quotes
"My only real hope left is to go with the pig heart, the xenotransplant." - Lawrence Faucette
"Mr Faucette's last wish was for us to make the most of what we have learned from our experience, so others may be guaranteed a chance for a new heart when a human organ is unavailable." - Dr. Bartley P. Griffith
Key Insights Distilled From
by Marilynn Lar... at www.medscape.com 11-01-2023
https://www.medscape.com/viewarticle/997985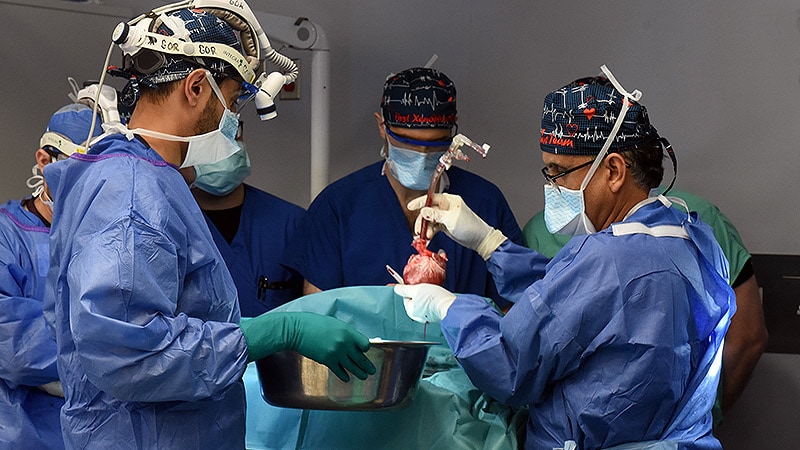
Deeper Inquiries
How can the medical community address the challenges of organ rejection in xenotransplantation?
Organ rejection in xenotransplantation can be addressed through various strategies. One approach is to further refine genetic modifications in pigs to make their organs more compatible with the human immune system. This could involve editing genes responsible for triggering immune responses or incorporating human genes into the pig genome to reduce rejection. Additionally, developing better immunosuppressive therapies tailored for xenotransplant recipients can help prevent rejection. Continuous monitoring of recipients for signs of rejection and prompt intervention are crucial in managing this challenge. Collaborative efforts between researchers, clinicians, and regulatory bodies are essential to address the complexities of organ rejection in xenotransplantation.
What ethical considerations arise from using genetically modified pig hearts for human transplants?
The use of genetically modified pig hearts for human transplants raises several ethical considerations. One major concern is the welfare of the animals involved in producing these organs. Ensuring that pigs are treated ethically and their genetic modifications do not cause unnecessary harm is crucial. There are also questions regarding informed consent and the decision-making process for patients considering xenotransplantation. Transparency about the risks, benefits, and uncertainties of this novel procedure is essential for upholding patient autonomy. Additionally, the allocation of these scarce resources and the potential implications for organ donation from deceased human donors need to be carefully considered from an ethical standpoint.
How can advancements in xenotransplantation research benefit other fields of medicine?
Advancements in xenotransplantation research have the potential to benefit various fields of medicine beyond organ transplantation. The development of genetically modified pigs with organs suitable for human transplantation can pave the way for personalized medicine approaches. Techniques used in xenotransplantation, such as gene editing and immunomodulation, can be applied to other areas of regenerative medicine and genetic therapies. Understanding the immune responses involved in xenotransplantation can also contribute to the treatment of autoimmune diseases and cancer immunotherapy. Moreover, the interdisciplinary nature of xenotransplantation research fosters collaboration between different medical specialties, leading to innovative solutions that can be translated to diverse healthcare challenges.
0
