Embolization Reduces Subdural Hematoma Surgery Need
The treatment of subdural hematomas has seen a significant advancement with the introduction of embolization procedures. Three randomized controlled trials, EMBOLISE, MAGIC-MT, and STEM, showcased the benefits of embolization in reducing the need for surgery and recurrence rates in patients with subdural hematomas. The trials utilized different embolization products, such as Onyx and Squid, to deliver the treatment. Embolization has emerged as a promising alternative to traditional surgical methods, offering improved outcomes and safety profiles for patients with subdural hematomas.
-
EMBOLISE Trial:
- MMA embolization with the Onyx product led to a significant reduction in the need for repeat surgical drainage of subdural hematomas.
- The embolization group showed lower rates of hematoma recurrence/progression and neurologic deterioration compared to the control group.
- Safety outcomes were favorable with minimal serious adverse events related to the embolization procedure.
-
MAGIC-MT Trial:
- The trial demonstrated that embolization was superior to usual care alone in reducing hematoma recurrence or progression.
- Functional outcomes were similar in embolization and control patients, with most patients showing good recovery.
- Serious adverse events were fewer in patients receiving embolization.
-
STEM Trial:
- Embolization therapy significantly reduced treatment failure rates in patients with chronic subdural hematomas.
- The procedure was beneficial for both surgical and nonsurgical patient populations, offering a promising treatment option.
- Safety outcomes were comparable between the standard management and embolization groups.
Customize Summary
Rewrite with AI
Generate Citations
Translate Source
To Another Language
Generate MindMap
from source content
Visit Source
www.medscape.com
Subdural Hematoma Embolization Success Cuts Surgery Need
Key Insights Distilled From
by at www.medscape.com 02-13-2024
https://www.medscape.com/viewarticle/subdural-hematoma-embolization-success-cuts-surgery-need-2024a100031u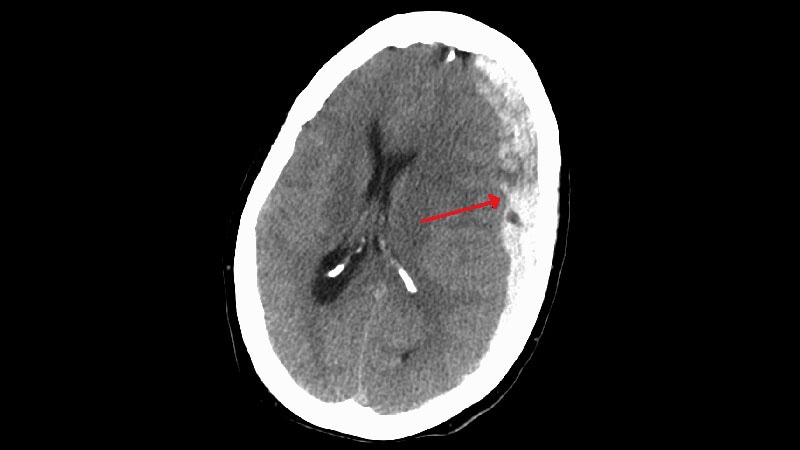
Deeper Inquiries
