Structural Insights into Bitter Taste Receptor TAS2R14 Activation by Cholesterol and an Intracellular Bitter Tastant
The article presents structural and functional insights into the bitter taste receptor TAS2R14, which is known to respond to a wide range of structurally diverse tastants. The key findings are:
-
The study reports two cryo-electron microscopy structures of TAS2R14 complexed with the G-proteins Ggust and Gi1. These structures reveal an orthosteric binding pocket occupied by the endogenous ligand cholesterol, as well as an intracellular allosteric site bound by the bitter tastant cmpd28.1.
-
Computational and biochemical analyses validate the binding of both cholesterol and cmpd28.1 to TAS2R14. Cholesterol is identified as an orthosteric agonist, while cmpd28.1 acts as a positive allosteric modulator with direct agonist activity.
-
The orthosteric pocket and allosteric site are connected via an elongated cavity with a hydrophobic core rich in aromatic residues, suggesting a potential mechanism for ligand recognition and signal transduction.
-
The findings provide insights into the diverse ligand recognition capabilities of bitter taste receptors, and suggest that TAS2R14 may have activities beyond bitter taste perception, potentially involving intracellular allosteric tastants.
Customize Summary
Rewrite with AI
Generate Citations
Translate Source
To Another Language
Generate MindMap
from source content
Visit Source
www.nature.com
Bitter taste receptor activation by cholesterol and an intracellular tastant - Nature
Key Insights Distilled From
by Yoojoong Kim... at www.nature.com 04-10-2024
https://www.nature.com/articles/s41586-024-07253-y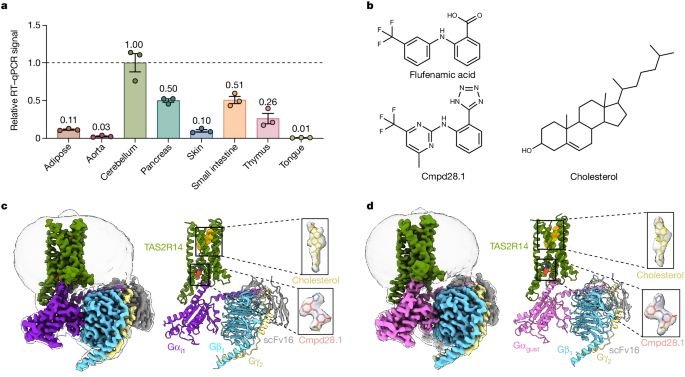
Deeper Inquiries
