Formation of Memory Assemblies Through the DNA-Sensing TLR9 Pathway
Core Concepts
Neurons undergo DNA damage and repair processes mediated by TLR9 during memory formation.
Abstract
The content discusses the formation of memory assemblies through the DNA-sensing TLR9 pathway in hippocampal neurons. It highlights the role of TLR9 in repairing DNA damage, inflammatory signaling, and memory formation processes. The content emphasizes the importance of maintaining TLR9 integrity for preventing neurocognitive deficits.
- Hippocampal neurons form memory microcircuits.
- Neurons undergo DNA damage and repair processes.
- TLR9 signaling is involved in memory formation.
- TLR9 plays a role in DNA damage repair and inflammatory responses.
- Implications of compromised TLR9 function on cognitive impairments and neurodegenerative disorders.
Customize Summary
Rewrite with AI
Generate Citations
Translate Source
To Another Language
Generate MindMap
from source content
Visit Source
www.nature.com
Formation of memory assemblies through the DNA-sensing TLR9 pathway - Nature
Stats
Neuron-specific knockdown of Tlr9 impaired memory.
TLR9 had an essential role in centrosome function.
TLR9-mediated repair results in recruitment to memory circuits.
Quotes
"Maintaining the integrity of TLR9 inflammatory signaling thus emerges as a promising preventive strategy for neurocognitive deficits."
Key Insights Distilled From
by Vladimir Jov... at www.nature.com 03-27-2024
https://www.nature.com/articles/s41586-024-07220-7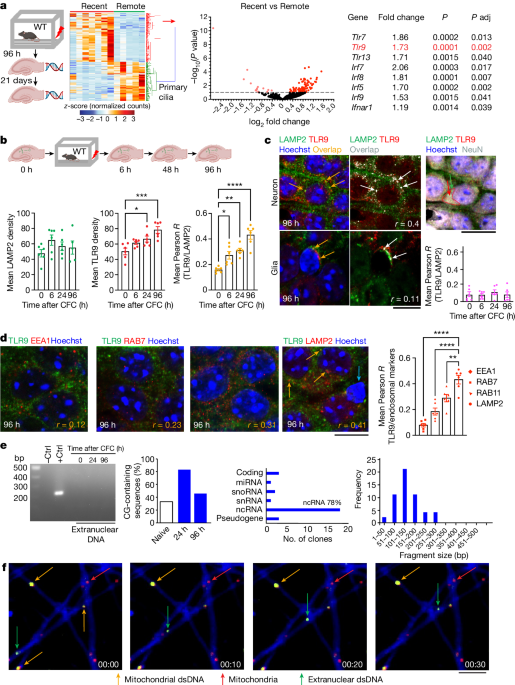
Deeper Inquiries
How does the role of TLR9 in memory formation impact potential therapeutic interventions
The role of TLR9 in memory formation presents a promising avenue for potential therapeutic interventions in cognitive impairments and neurodegenerative disorders. The study highlights how TLR9 signalling is crucial in the formation of memory assemblies in hippocampal neurons, with its activation leading to the repair of DNA damage and the recruitment of neurons to memory circuits. By understanding the intricate molecular events involved in TLR9-mediated memory formation, researchers can explore targeted interventions that enhance TLR9 function. This could involve developing drugs or treatments that modulate TLR9 signalling to improve memory formation and cognitive function. Additionally, maintaining the integrity of TLR9 inflammatory signalling emerges as a preventive strategy for neurocognitive deficits, suggesting that therapies aimed at preserving TLR9 function could potentially mitigate cognitive decline and memory impairments in various neurological conditions.
What are the implications of compromised TLR9 function on genomic stability
Compromised TLR9 function can have significant implications on genomic stability, leading to a cascade of molecular events that may contribute to cognitive impairments and neurodegenerative disorders. The study reveals that neurons with persistent dsDNA breaks and nuclear envelope ruptures undergo TLR9-mediated repair processes to maintain genomic integrity. When TLR9 function is impaired, these repair mechanisms may be compromised, resulting in unresolved DNA damage and genomic instability in neurons. This instability can lead to accelerated senescence, psychiatric disorders, and neurodegenerative conditions. Therefore, compromised TLR9 function not only affects memory formation but also poses a risk to genomic stability, potentially exacerbating cognitive impairments and neurodegenerative processes.
How can the understanding of DNA damage and repair mechanisms in neurons contribute to advancements in neurodegenerative disorder treatments
The understanding of DNA damage and repair mechanisms in neurons offers valuable insights that can contribute to advancements in neurodegenerative disorder treatments. The study demonstrates a novel cascade of molecular events in hippocampal neurons undergoing dsDNA damage and TLR9-mediated repair, which are essential for memory formation. By elucidating these mechanisms, researchers can develop targeted therapies that enhance DNA repair processes in neurons, potentially slowing down the progression of neurodegenerative disorders. Strategies aimed at preserving genomic stability and promoting efficient DNA damage repair, such as modulating TLR9 signalling, could offer new therapeutic approaches for conditions like Alzheimer's disease, Parkinson's disease, and other neurodegenerative disorders. This deeper understanding of DNA damage and repair mechanisms in neurons opens up avenues for developing innovative treatments that target the molecular processes underlying these debilitating conditions.
0
